Example of a primary drying design space graph showing sublimation
By A Mystery Man Writer

A Software Tool for Lyophilization Primary Drying Process Development and Scale-up Including Process Heterogeneity, I: Laboratory-Scale Model Testing

Technical transfer and commercialisation of lyophilised biopharmaceuticals — application of lyophiliser characterisation and comparability, AAPS Open

Figure A1. Product temperature for 5.4 mL and 10.6 mL fill volumes

Processes, Free Full-Text

Schematic evolution of a Freeze-drying cycle. Adapted from

Tong ZHU, Senior Scientist I, Ph.D. in Aero Eng.

PDF) Recommended Best Practices for Lyophilization Validation—2021
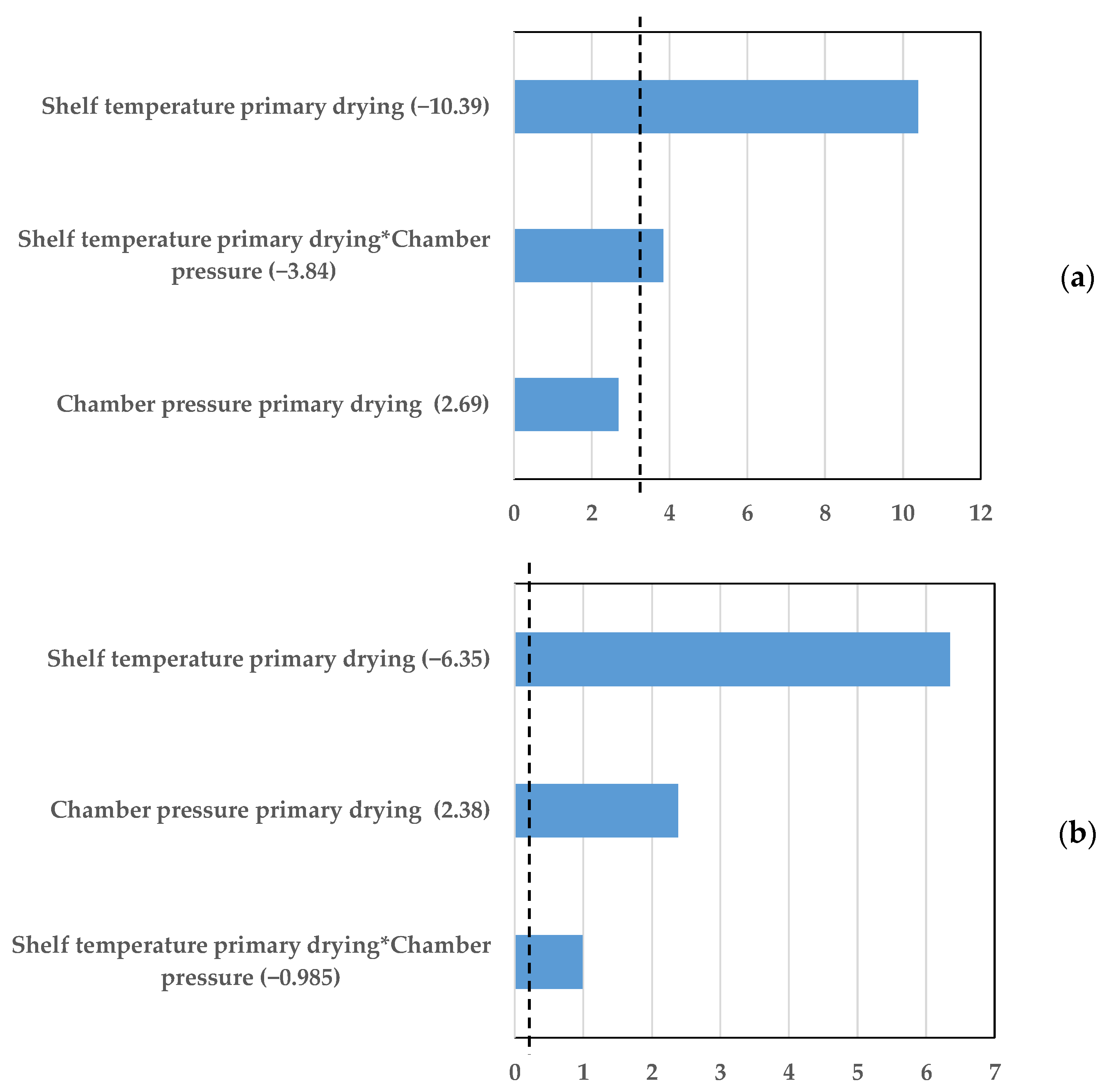
Processes, Free Full-Text

Lyophilization Process Design Space - ScienceDirect

Freeze-drying parameters for uncontrolled and controlled

The graphical design space for the primary drying phase of freeze Drying: Factors affecting the dried product layer resistance - ScienceDirect

Full article: Integrated use of mechanistic models and targeted experiments for development, scale-up and optimization of lyophilization cycles: A single vial approach for primary drying
- Graphic - Custom Soccer Jerseys Kit Sublimated for Academy-XTeamwear

- 4 Free Blank Pink Sublimation Graphics - Free Pretty Things For You

- Pattern- Sublimation T-Shirt Printing Services golden graphics

- What Type of Shirt is Best for Sublimation? — dasFlow

- Sublimation for Beginners: A Step-by-Step Tutorial - Cranky Press Man

- Hanes Men's Tagless ComfortFlex Waistband, Multi-Packs Available Brief, 3-pack, Medium at Men's Clothing store: Briefs Underwear

- Golden hour wearing @spanx 🌅💛 Use my promo code - ACTIVEASHLEYG for 15% off select activewear to shop. Link in bio! #SpanxPartner
- Athletic Seamless Sports Bra (Lilac) – Fitness Fashioness

- Tops To Wear With Brown Pants International Society of Precision Agriculture

- Eye Candy': TV Review

