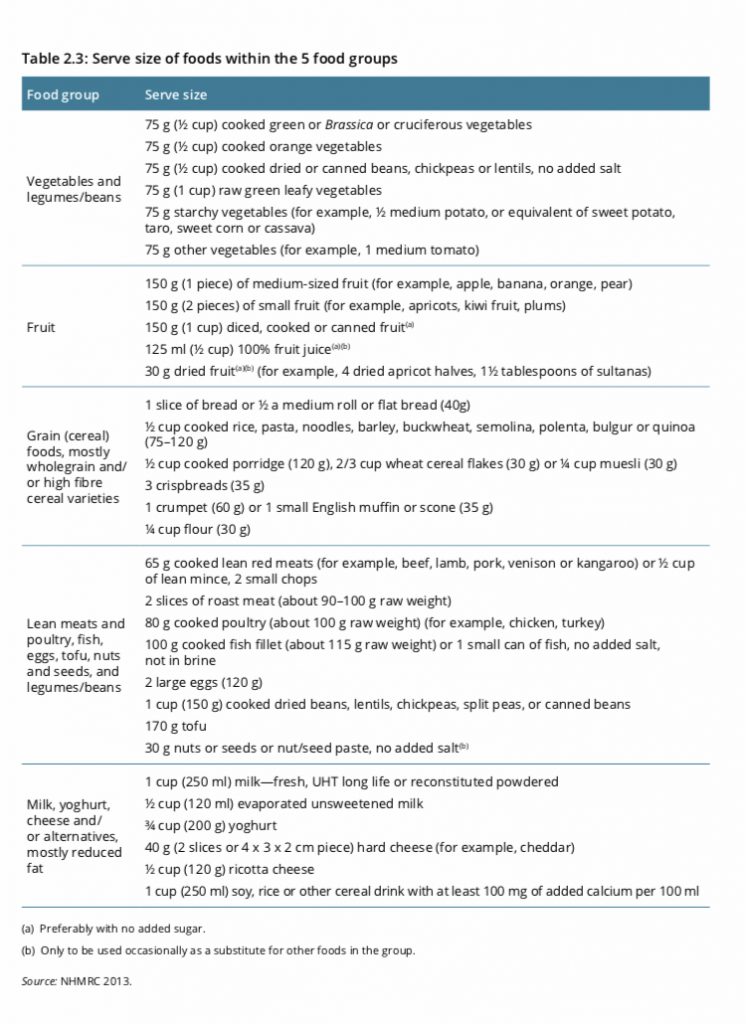
Solved Problem 3 (Phase changes) A 35 g ice cube at 0°C is

By A Mystery Man Writer
Answer to Solved Problem 3 (Phase changes) A 35 g ice cube at 0°C is
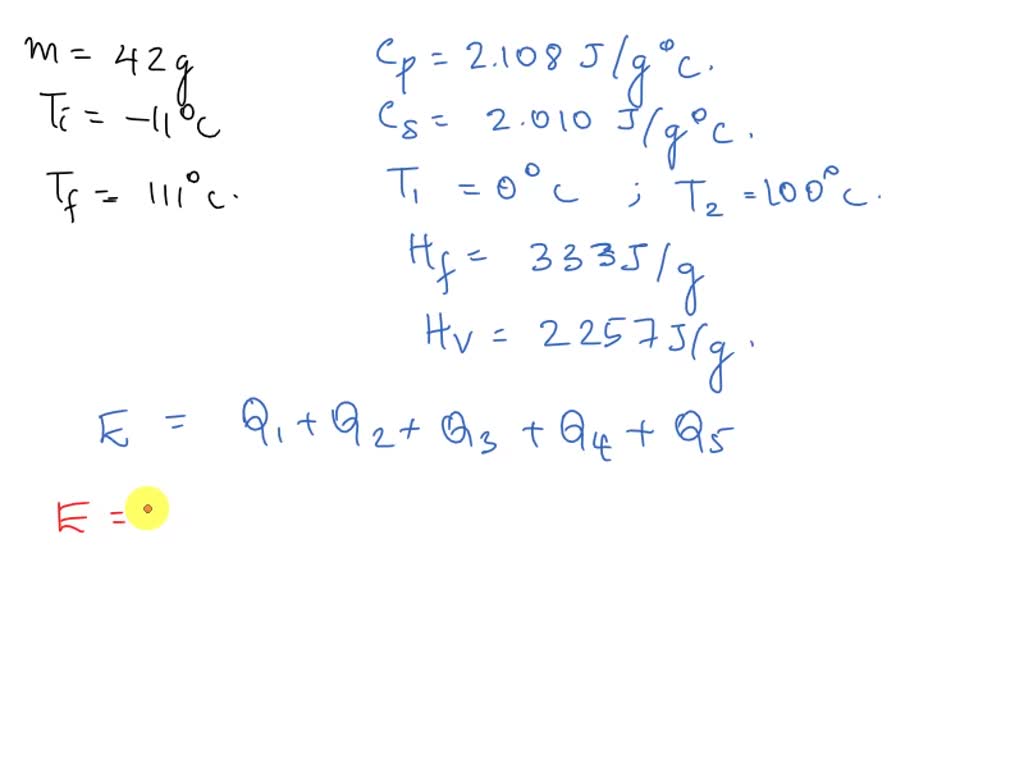
SOLVED: How much energy is required to change a 42g ice cube from ice at -11c to steam at 111c

Thermochemistry: Water Phase Change Heat Calculation.wmv

14.3 Phase Change and Latent Heat

SOLVED: A 35 g ice cube at 0.0 °C is added to 110 g of water in a 62 g aluminum cup. The cup and the water have an initial temperature of

Hard brain teaser #29
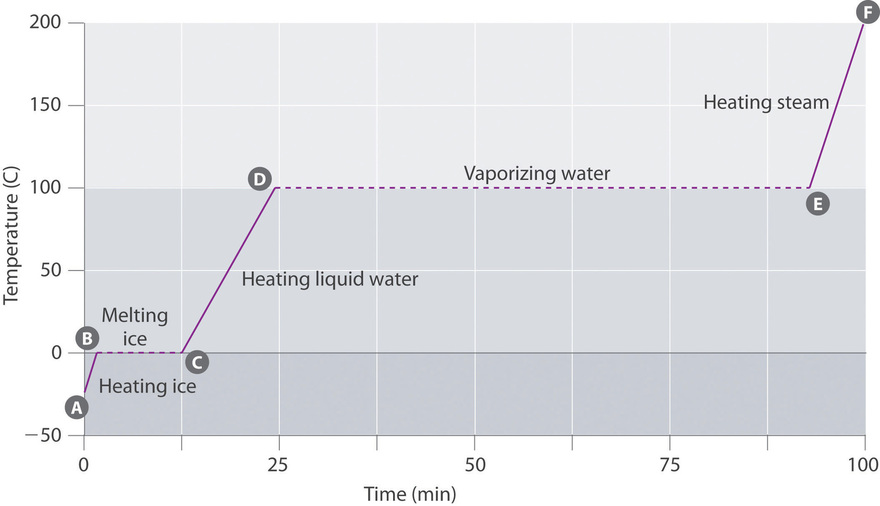
11.6: Phase Changes - Chemistry LibreTexts

Quantum gravity - Wikipedia
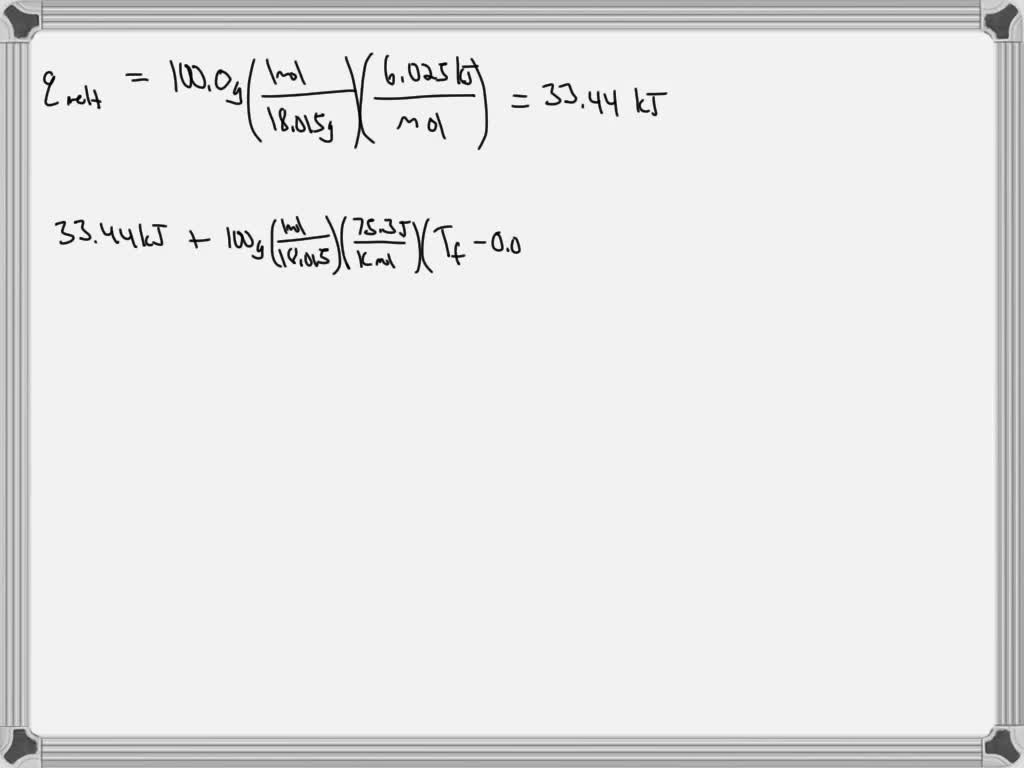
SOLVED: An ice cube at 0 °C weighing 100.0 g is dropped into kg of water at 20 °C. Does all of the ice melt? If not, how much of it remains?

14.3 Phase Change and Latent Heat – College Physics: OpenStax

Solved If the student had instead added 59 g of ice

Core-sheath phase change fibers via coaxial wet spinning for solar energy active storage - ScienceDirect
- 3D Porsche Mission X Concept 2023 - TurboSquid 2122795

- Pantalones cortos de verano con cintura elástica para mujer, con bolsillos, cintura alta, volantes

- FASHIONWT Women Plus Size Hips Sponge Pad Fake Buttocks Sexy Hip Pad Beautiful Butt Body Shaping Pants

- Shop Bonds Men's Guyfront Trunks 3-Pack - Black/Metallic Gold

- Annadress Women's A Line Prom Dresses Spaghetti Straps Evening Gowns Prom Party Gowns Black 2 : : Clothing, Shoes & Accessories