000559 Calculation of Compressibility Factor from Redlich-Kwong Equation

By A Mystery Man Writer

SOLVED: The van der Waals and Redlich-Kwong Equations, which are used in high-pressure conditions, are as follows: The van der Waals equation: (P + a(n/V)^2)(V - nb) = nRT where: P

Variation of properties in the dense phase region; Part 2 – Natural Gas

Solved] The Redlich-Kwong equation of state is gi
Compressibility Factor Calculator - Community

Solved] The Redlich-Kwong equation of state is gi

Ideal Gas, Van der Waals, and Redlich-Kwong Equations of State with Compressibility

Compressibility Factors Using the Soave-Redlich-Kwong Equation of State - Wolfram Demonstrations Project

Compressibility Factors Using the Soave-Redlich-Kwong Equation of State - Wolfram Demonstrations Project
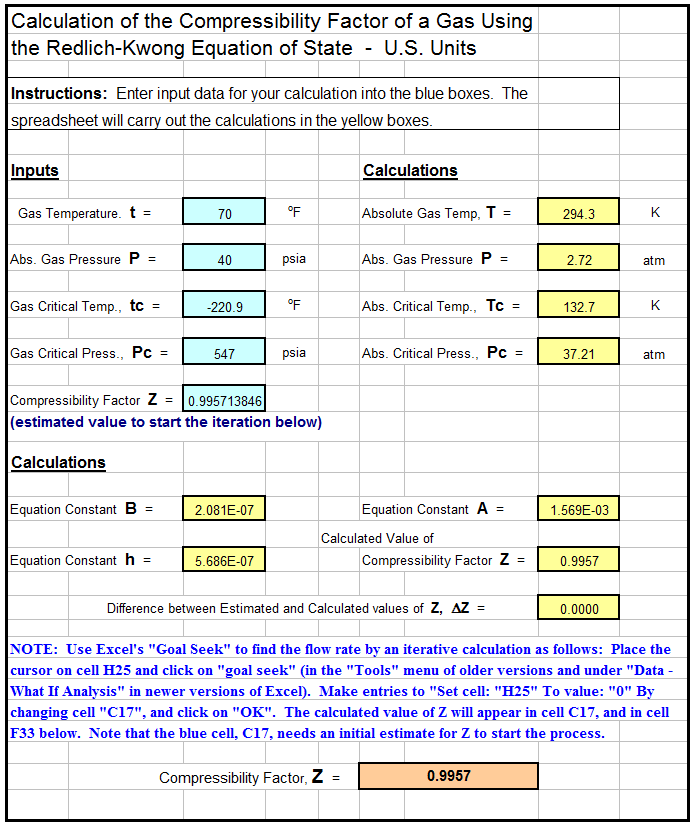
Redlich Kwong Archives - Low Cost Easy to Use Spreadsheets for Engineering Calculations Available at Engineering Excel SpreadsheetsLow Cost Easy to Use Spreadsheets for Engineering Calculations Available at Engineering Excel Spreadsheets

Solved The Redlich-Kwong equation of state for a gas is P=

Redlich–Kwong equation of state - Wikipedia

2. (a) Derive an expression for the isothermal
- KILZ 2® ALL-PURPOSE Interior/Exterior Primer

- Tostones de Pana - Puerto Rican Cuisine

- Full Back Coverage Bras For Women, Fashion Deep Cup Hide Back Fat Bra With Shapewear Incorporated Push Up Sports Bras

- Plus Size Tek Gear® Basic Capri Bottom clothes, Plus size, Clothes

- Prettylittlething Pink Sport Textured Booty Shorts




